Category: Joe Blogs
From Samples to Insight: Meet the New Agilent Cytation 9
A Joe Blogs post by Joe Roberts, PhD
As biological models become more complex – from 3D cultures to longitudinal live-cell assays – the real bottleneck in research isn’t data generation. It’s integration. Imaging, quantitation, kinetics… too often these sit in separate workflows, separate instruments, and separate datasets.
The newly released Agilent Cytation 9 Cell Imaging Microplate Reader from Agilent Technologies takes a different approach: one platform, multiple data dimensions, and a clear path from samples to actionable insight.
Agilent Cytation 9: One Platform, Two Microscopes, Endless Applications

At the heart of Cytation 9 is something genuinely distinctive — it combines multimode plate reading with both inverted and upright microscopy in a single system.
This dual-microscope configuration opens up a broader experimental space:
- Inverted imaging for standard cell-based assays
- Upright imaging for applications like slides, ELISpot, or tissue sections
- Seamless switching between modalities without moving samples
It’s not just consolidation – it’s capability expansion.
Designed to Extract More Data Per Experiment
Cytation 9 is clearly built around maximising information density.
Key enhancements include:
- Expanded image cube capacity to capture more fluorescence channels per well
- High-resolution APO objectives for improved image quality
- Wide range of imaging modes, including fluorescence, brightfield, phase contrast, and multi-colour imaging
The result? Richer datasets from every experiment – without increasing sample volume or complexity.
Speed and Throughput Without Compromise
One of the most practical upgrades is throughput.
Cytation 9 delivers:
- Faster imaging workflows to move from acquisition to analysis more efficiently
- Automated features like laser autofocus and montage imaging
- Support for high-density formats (up to 1536-well plates)
Combined, these improvements help labs scale from routine assays to higher-throughput screening without rethinking their entire workflow.
Built for Live-Cell and Kinetic Workflows
Where Cytation 9 really comes into its own is in dynamic biology.
With software-controlled environmental regulation, including CO₂ and O₂ control, temperature, and humidity, the system enables stable long-term live-cell imaging.
This allows researchers to:
- Track cellular responses over time
- Run kinetic assays with confidence
- Reduce variability introduced by external incubation systems
It’s a shift from static snapshots to continuous biological insight.
Flexibility That Grows With Your Lab
Cytation 9 isn’t a fixed system – it’s modular.
You can:
- Add imaging modes as your applications evolve
- Integrate with automation platforms like BioSpa™ or BioStack™
- Work across diverse labware, from plates to flasks and slides
This makes it equally relevant for core facilities, translational labs, and drug discovery environments where needs change rapidly.
Joe’s Takeaway
The Cytation 9 stands out because it doesn’t force a trade-off between imaging and quantitation – it integrates both in a way that actually improves experimental design.
The addition of upright microscopy alongside the traditional inverted setup is a particularly smart move. It broadens what the system can do without adding complexity, and that’s ultimately what most labs need: more capability, not more workflow friction.
If your work involves cell-based assays, imaging, or high-content workflows, Cytation 9 is well worth a closer look.
If you’re interested in a demo or exploring how this could fit into your lab, feel free to contact us today.
Until next time… happy experimenting!

On this page
Why IFN-γ Remains One of the Most Measured Cytokines in Immunology Research
A Joe Blogs post by Joe Roberts, PhD
If you spend any time studying immune responses, you’ll quickly encounter interferon gamma (IFN-γ). It’s one of the most widely measured cytokines in immunology research, and at Millennium Science, it’s also one of the most frequently ordered reagents in our catalogue.
That’s hardly surprising. IFN-γ sits at the centre of many immune processes and is widely used as a functional readout of cellular immune activity. From infectious disease research to cancer immunology and vaccine development, measuring IFN-γ often provides a direct window into how immune cells are responding.
Let’s take a closer look at why this cytokine is so important, and the different ways researchers are measuring it today.

What is IFN-γ?
Interferon-γ (IFN-γ) is a type II interferon primarily produced by:
- CD4⁺ Th1 cells
- CD8⁺ cytotoxic T cells
- Natural killer (NK) cells
Unlike type I interferons (such as IFN-α and IFN-β), which are produced by many cell types, IFN-γ is mainly produced by activated immune cells during adaptive and innate immune responses.
Functionally, IFN-γ contributes to several important immune processes:
- Antiviral immunity – enhances immune responses against infected cells
- Immunomodulation – upregulates antigen presentation through increased MHC expression
- Macrophage activation – a key signal driving classical macrophage activation
- Pro-inflammatory signalling – promotes Th1-type immune responses
Because of these roles, IFN-γ is frequently used as a readout of cellular immune activation, particularly when studying T-cell responses.
Why Do Researchers Measure IFN-γ So Often?
In many immune assays, IFN-γ acts as a functional marker of antigen-specific immune activity.
Researchers often measure IFN-γ to:
- Evaluate vaccine-induced immune responses
- Assess T-cell immunity to infection
- Study tumour-specific immune responses
- Characterise Th1-biased immune signalling
- Monitor cell therapy potency
This helps explain why IFN-γ assays remain among the most requested cytokine reagents in our portfolio.
Mabtech Offers Four Ways to Measure IFN-γ
Different experiments require different levels of sensitivity, multiplexing, or single-cell resolution. Fortunately, IFN-γ can be detected using several complementary technologies.
- ELISA – Quantifying Secreted IFN-γ
ELISA remains one of the most widely used cytokine detection methods.
It provides:
- Quantitative measurement of IFN-γ in supernatants, serum, or plasma
- High sensitivity and reproducibility
- A straightforward workflow compatible with most labs
ELISA is ideal when you want to measure total cytokine secretion from a population of cells.
- ELISpot – Detecting IFN-γ at the Single-Cell Level
ELISpot takes cytokine detection one step further by measuring secretion from individual cells.
This technique enables researchers to:
- Quantify IFN-γ-secreting cells
- Detect rare antigen-specific T cells
- Achieve exceptional sensitivity
Because of this sensitivity, ELISpot has become a widely used approach for T-cell immune monitoring, particularly in vaccine and immunotherapy research.
- FluoroSpot – Multiplex Cytokine Detection
FluoroSpot expands on ELISpot by allowing simultaneous detection of multiple cytokines using fluorescent detection.
This makes it possible to identify polyfunctional immune cells, which can provide deeper insights into immune responses.
For example, researchers can detect:
- Triple-secreting IFN-γ / IL-2 / TNF-α T cells
- Killer cells secreting IFN-γ and Granzyme B
This type of analysis provides a much richer picture of immune responses, especially when studying complex cell populations.
- EYRAPlex – Multiplex Cytokine Quantification
For researchers who want high-throughput multiplex cytokine profiling, EYRAPlex assays provide a powerful alternative.
EYRAPlex enables:
- Simultaneous measurement of multiple cytokines
- High-throughput cytokine profiling
- Compatibility with the EYRA instrument platform and most Flow Cytometers
This approach is ideal when researchers want to explore complex cytokine signatures rather than focusing on a single immune marker.
EOFY Promotion: 10% Off IFN-γ FluoroSpot Assays
Because IFN-γ remains such a critical immune readout, we’re currently running a limited EOFY promotion.
Until 30 June, you can receive: 10% off Mabtech IFN-γ FluoroSpot assays
If you’re already measuring IFN-γ responses, or planning a new T-cell study, it’s a great opportunity to expand your toolkit.
Final Thoughts
From infection biology to immunotherapy, IFN-γ remains one of the most informative cytokines to measure when studying cellular immunity.
Whether you’re quantifying cytokine secretion with ELISA, detecting rare antigen-specific cells with ELISpot, studying polyfunctional responses with FluoroSpot, or analysing complex cytokine networks with EYRAPlex, there are now multiple powerful tools available to explore IFN-γ biology.
If you’d like more information about IFN-γ assays or the current EOFY promotion, feel free to reach out.

Until next time… happy experimenting!
Seeing More, Doing More: Agilent’s BioTek Imaging Portfolio in Action!
A Joe Blogs post by Joe Roberts, PhD
Modern life science research increasingly demands more information from fewer samples, delivered faster and with greater confidence. Whether you’re studying immune cell function, complex 3D models, or subtle phenotypic changes, imaging is central to generating meaningful insights. Agilent’s BioTek imaging portfolio is designed to meet this challenge, combining high-quality microscopy, flexible multimode detection, and live-cell capability into integrated platforms that scale with your research needs.
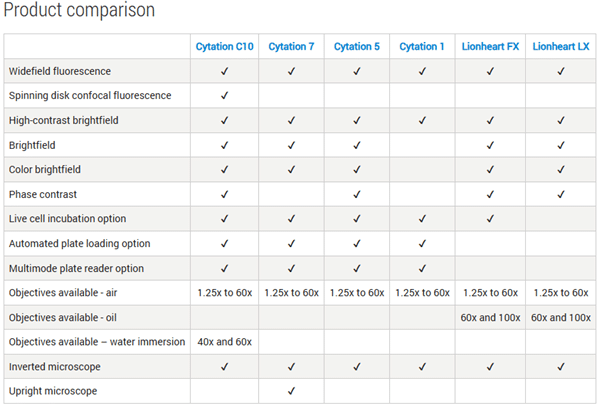
Cytation and Lionheart: Imaging Platforms That Scale With Your Research
The Agilent BioTek Cytation™ range spans entry-level widefield imaging through to advanced confocal workflows, combining automated microscopy with multimode microplate reading in a single, integrated platform. Designed to scale with experimental complexity, Cytation systems support routine plate-based assays, high-content imaging, and live-cell experiments with environmental control, making them suitable for laboratories of all sizes and budgets.
For labs requiring dedicated automated microscopy, the Agilent BioTek Lionheart™ FX and LX microscopes deliver high-performance imaging in a compact footprint. These systems support a wide range of objectives and imaging modalities, with robust autofocus, flexible incubation options, and automation-ready workflows. Lionheart platforms are particularly well suited to phenotypic profiling, kinetic assays, and live-cell imaging where precision and repeatability are essential.
A detailed product comparison table below highlights the specific capabilities of each Cytation and Lionheart model.
Why Agilent BioTek Imaging Stands Out
Across the portfolio, Agilent imaging platforms share several key advantages:
- Support for 6- to 1536-well plates, slides, dishes, and flasks.
- Compatibility with automation solutions like BioStack™, BioSpa™, and BenchCel™.
- Live-cell imaging with CO₂/O₂ control, shaking, and temperature incubation.
- Advanced imaging capabilities: Z-stacking, Z-projection, montage stitching, and time-lapse videos.
- Hybrid optics for sensitive and specific fluorescence detection.
Applications include wound healing assays, cell migration and invasion studies, immunofluorescence, neurite outgrowth analysis, 3D cell models, and ADME/Tox experiments. By unifying imaging and detection in a single platform, Agilent BioTek systems help labs move beyond single-endpoint measurements to richer, biologically meaningful data.
Joe’s Takeaway
Whether you’re starting with simple plate-based assays or pushing the boundaries of confocal or automated microscopy, the Agilent BioTek imaging portfolio gives you the flexibility, performance, and scalability to see more and do more.
If you’re interested in a demo, contact us today!
Until next time… happy experimenting!

One Year In: Reflections on Life, Science and Supporting Discovery at Millennium Science
A Joe Blogs post by Joe Roberts, PhD
It’s hard to believe it’s already been one year since I joined Millennium Science. Over the past 12 months, I’ve had the privilege of working closely with researchers across Australia and New Zealand, supporting everything from early-stage assay development through to established, high-throughput workflows. It’s been a year full of learning, connection, and a renewed appreciation for the science happening right on our doorstep.

A Front-Row Seat to Great Science
One of the biggest positives of this role has been the variety. No two weeks look the same, from visiting labs and running demos, to attending conferences and catching up with researchers over morning teas. Those conversations, whether at the bench or over a coffee, often provide the most valuable insights into what researchers actually need.
Events like CYTO-Connect and ASI have been particular highlights. There’s something energising about seeing the community come together, sharing data, debating approaches, and learning from one another. Being able to support these meetings, demonstrate new technologies, and have real, practical conversations about workflows has been incredibly rewarding.
From the Bench to the Field
Coming into this role from a background as a Post-Doctoral Research Fellow, I initially saw the move as an opportunity to apply my scientific training in a different way. What I’ve learned is just how valuable that bench-side experience is when supporting researchers, understanding experimental constraints, timelines, and the realities of generating reliable data.
Over the past year, that experience has shaped how I work: launching and writing Joe Blogs to share practical insights from the field, hosting webinars to break down new technologies, providing hands-on instrument and assay demos, and presenting at national conferences. Across all of these, the goal has remained the same, to translate complex technologies into something genuinely useful for real research questions.
What the First Year Has Taught Me
This past year has also been a steep (and enjoyable) learning curve. A few key takeaways stand out:
- No two labs are the same – listening is always more valuable than assuming.
- Technology is only powerful when it’s accessible – training, support, and context matter just as much as specifications.
- Collaboration drives outcomes – the best results come from strong partnerships between researchers, suppliers, and local support teams.
Science Is Serious – But Culture Matters Too
At Millennium Science, science is serious – but culture matters just as much. It’s not just about cutting-edge tech; it’s about curiosity, collaboration, and actually enjoying the workday. We’ve got a dog-friendly office, share knowledge across teams, and even throw epic end-of-year battle cart competitions (which Joe Blogs won last year).
That mix of expertise, energy, and a little friendly fun makes it a joy to help researchers tackle real-world challenges, turning complex tech into solutions that actually make a difference in the lab.
Looking Ahead
As I head into year two, I’m excited to continue supporting researchers through lab visits, demos, webinars, conferences, and everything in between. If you’re planning a new project, exploring a new technology, or just want to talk through an idea over a coffee, I’d love to hear from you (contact us today).

Until next time… happy experimenting!

Unleashing Spectral Power: Agilent NovoCyte Opteon Arrives in ANZ
A Joe Blogs post by Joe Roberts, PhD
Spectral flow cytometry continues to transform single-cell research – and now, for the first time, laboratories in Australia and New Zealand can experience the Agilent NovoCyte Opteon first hand.
Launched globally at CYTO 2024, the NovoCyte Opteon has already earned international recognition, taking home the Select Science Scientist’s Choice Award for “Best New Drug Discovery & Development Product of 2024.” With up to five lasers, 73 detectors, and cutting-edge optical engineering, this system represents the next generation of high-dimensional cytometry – and it’s now available for demo in ANZ.
Spectral acquisition, redefined
Unlike conventional cytometry, the NovoCyte Opteon captures the full emission spectra of each fluorochrome across all lasers. This approach allows for greater panel flexibility, more accurate unmixing, and cleaner resolution of overlapping signals – ideal for complex immunophenotyping studies.
Spectral Flow Cytometry vs Conventional Flow: What’s the Difference?
Traditional flow cytometry detects fluorescence using individual optical filters for each fluorochrome, limiting panel size and often causing signal overlap.
Spectral flow cytometry – like the Agilent NovoCyte Opteon – captures the entire emission spectrum from every fluorochrome across all lasers.
This enables:
-
Greater panel flexibility with more markers in a single run
-
Improved accuracy through spectral unmixing of overlapping signals
-
Cleaner data by accounting for autofluorescence as a separate spectral component
The result is a more powerful, precise, and reproducible analysis – ideal for complex immunophenotyping and high-dimensional research.
Configurability and Optics
Researchers can choose 3-, 4-, or 5-laser configurations, with the flagship system spanning UV, Violet, Blue, Yellow/Green, and Red lasers – and up to 73 detectors. Agilent’s proprietary optics and electronics maximise sensitivity and spectral separation, ensuring high-quality data.
Dynamic Range, Small-Particle Detection, and Autofluorescence Handling
It boasts a wide dynamic range both for fluorescence and scatter (size) detection, reducing the need for frequent detector adjustments.
Dual-laser small particle detection (using 405 nm and 488 nm SSC) enables detection of particles down to ~80 nm without needing separate adjustments between cell and particle modes.
Also, the instrument supports autofluorescence subtraction (i.e. treating autofluorescence as a spectral component), which helps resolve dim populations more clearly.
Reliability and Stability Built In
To maintain performance in variable lab environments, the NovoCyte Opteon integrates on-board temperature control, fluidics monitoring, electronics sensor circuits, and real-time instrument status feedback.
Automation and Throughput
It’s compatible with the NovoSampler S, accepting 40-tube racks and microplates (384/96/48/24), and is ready for robotic automation. Calibration is automated, with templates for labware types saved for reproducibility. Carryover is minimal (< 0.1 %) via rinse cycles.
Software and Workflows
Agilent’s NovoExpress (Opteon) software version 2.0+ underpins the acquisition, unmixing, analysis, and reporting workflow. They’ve enhanced the user interface with an “unmixing” tab, streamlining spectral unmixing steps. The software supports both real-time acquisition and downstream “offline” analyses.
Joe’s Takeaway
The arrival of the Agilent NovoCyte Opteon in the ANZ region marks a real milestone for spectral flow cytometry. Local researchers now have access to one of the most advanced, award-winning platforms available – combining powerful optics, automation readiness, and Agilent’s renowned reliability, all supported locally by Millennium Science.
If you’re interested in demoing the award-winning NovoCyte Opteon Spectral Flow Cytometer, we’d love to hear from you. Reach out to arrange a hands-on session and see what spectral flow can really do for your research.
And if you’re attending CYTO-Connect (Perth, November 27-29, 2025), come and say hello – we’ll be there showcasing the Opteon and chatting all things spectral!
If you’re interested in a demo of the Opteon, contact us today!
Until next time… happy experimenting!

On this page
- A Joe Blogs post by Joe Roberts, PhD
- Spectral acquisition, redefined
- Spectral Flow Cytometry vs Conventional Flow: What’s the Difference?
- Configurability and Optics
- Dynamic Range, Small-Particle Detection, and Autofluorescence Handling
- Reliability and Stability Built In
- Automation and Throughput
- Software and Workflows
- Joe’s Takeaway
Real-Time Cell Insights with Agilent xCELLigence RTCA eSight
A Joe Blogs post by Joe Roberts, PhD
Cells don’t stand still, so why should your assays? The Agilent xCELLigence RTCA eSight™ goes beyond snapshots, revealing real-time cell behaviour with label-free impedance and live imaging.
By pairing biosensor impedance technology with live-cell imaging, eSight enables researchers to monitor cell health, function, and behaviour simultaneously, continuously, and in real time. It’s not just more data, it’s deeper, more meaningful insight into what your cells are really doing.

How Real Time Cell Analysis (RTCA) Technology Works
At the heart of RTCA eSight are proprietary E-Plates, embedded with gold biosensors. These electrodes non-invasively measure impedance, which reflects cell metrics such as:
- Proliferation
- Adhesion strength
- Morphological changes
- Migration and differentiation
Because impedance is recorded continuously, you capture events as they happen, in seconds, minutes, hours, or days, without disturbing the cells. The temporal resolution is exquisite, allowing you to see subtle shifts long before they’re visible under a microscope.

Multi-Modal Cell Analysis: Impedance Meets Live-Cell Imaging
In concert with impedance, eSight’s imaging module provides brightfield plus three fluorescence channels (red, green, blue). This lets you visualise your cells directly while validating and enriching the kinetic impedance data.
The result is a spatial and temporal view of cell populations at an unprecedented level of detail, ideal for assays like proliferation, cytotoxicity, and apoptosis. And importantly, impedance and imaging are performed on the same cells, not replicate wells, so your data are directly correlated.
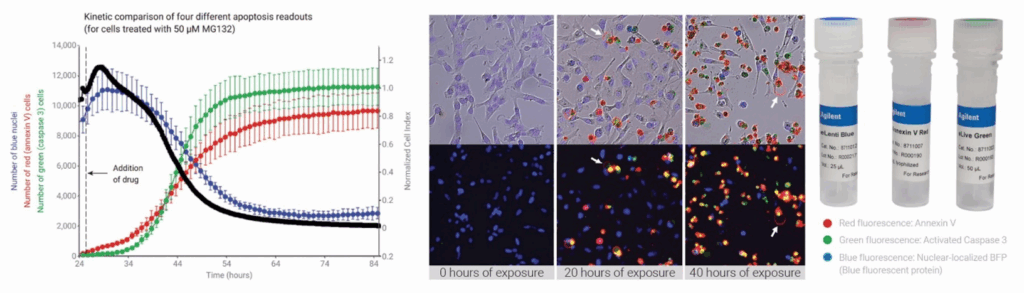
Streamlined RTCA Workflow
One setup, two data streams. Here’s how simple it is:
- Seed your cells into an E-Plate.
- Insert the plate into eSight (inside your incubator).
- Define your assay in the RTCA software.
From there, impedance and imaging data are captured automatically, then integrated into a single timeline for straightforward analysis. The software even supports outputs like:
- RTCA images
- KT50 (time to 50% cytolysis at a given effector-to-target ratio)
- % Cytolysis dose response curves
- IC50 calculations
No juggling between platforms – just a unified dataset ready for export.

Applications Across the Board
Researchers are already applying RTCA eSight to:
- Immune-cell killing assays – track cytolysis in real time with kinetic precision.
- Virology – Screening and characterizing antiviral drugs in real time
- Proliferation and apoptosis studies – capture early events, validate with imaging, and overlay with fluorescent markers.
- Cell heterogeneity analysis – reveal subpopulation responses that would be lost in averaged data.

Joe’s Takeaway
The Agilent xCELLigence RTCA eSight is more than the sum of its parts. By integrating patented biosensor impedance with live-cell imaging, it delivers:
- Continuous, label-free monitoring of cell health and function
- Brightfield and Three-colour live-cell imaging for direct visual validation
- Unified kinetic datasets from the same cell population
- Broad versatility across immuno-oncology, virology, and general cell biology
For researchers who want to move beyond static snapshots, eSight offers a powerful new way to see biology unfold in real time, with the reproducibility, temporal resolution, and ease-of-use that modern labs demand.
If you’re interested in a demo of the eSight, contact us today!
Until next time… happy experimenting!

Meet EYRA: Multiplexing Reimagined
A Joe Blogs post by Joe Roberts, PhD
When it comes to multiplex protein analysis, researchers need accuracy, speed, and simplicity. Traditional flow-based systems have long been the standard, but they bring challenges: sheath fluids, blocked probes, and constant maintenance. Enter the Mabtech EYRA™ – a fluidics-free multiplex immunoassay platform that reimagines how scientists generate cytokine and biomarker data. With confocal imaging, RAWsphere analysis, and compatibility with EYRAplex bead kits, EYRA makes multiplexing faster, simpler, and more reliable.


Multiplex Without Compromise
With EYRAplex magnetic bead assays, EYRA can quantify more than 30 analytes from a single sample – whether that’s serum, plasma, or cell culture supernatant. This means less sample consumption, fewer runs, and richer datasets for every experiment.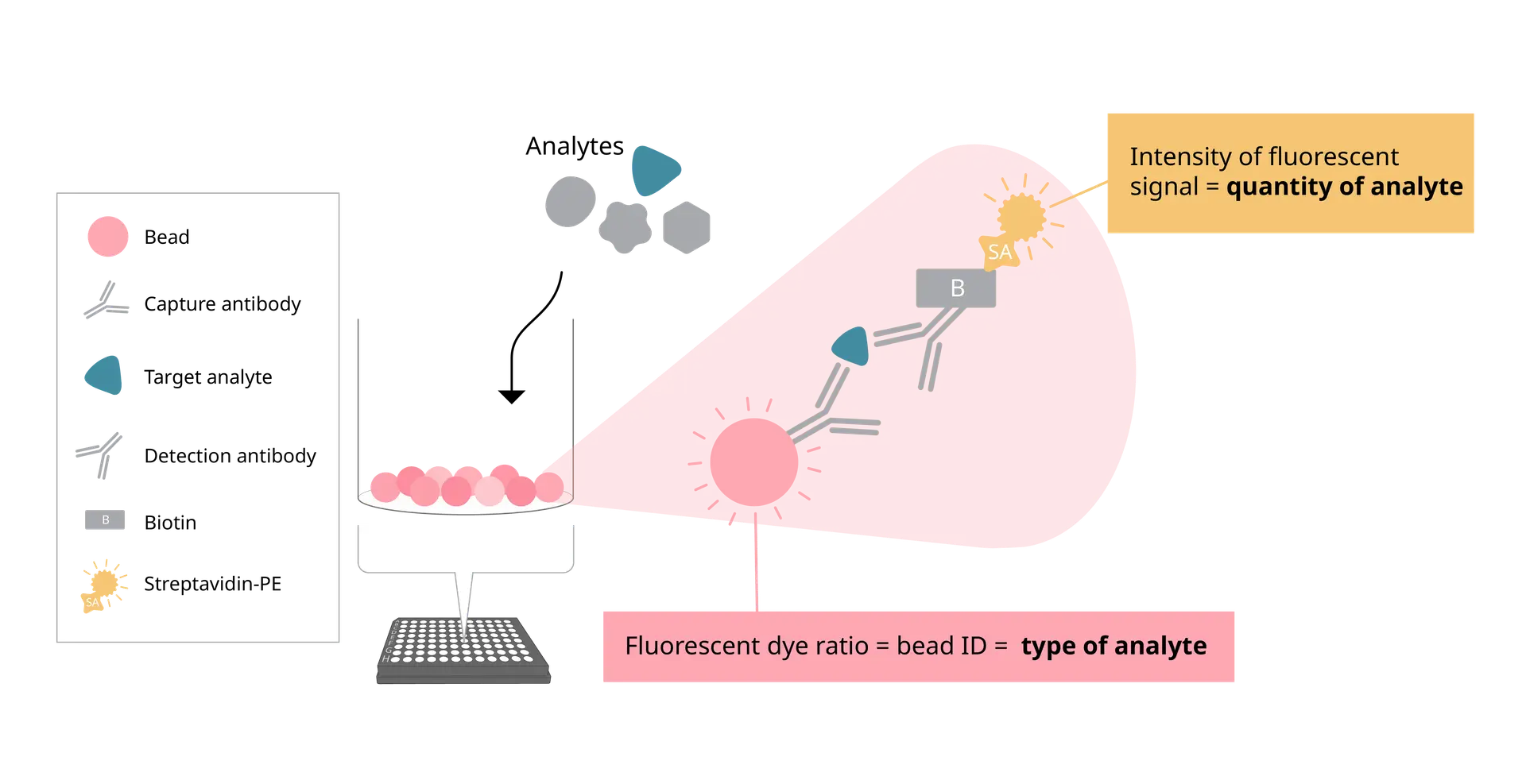
At the core of EYRA’s precision is the RAWsphere image analysis algorithm. It identifies each bead, links it to the correct analyte, and quantifies the PE signal with high resolution. Whether you’re measuring six cytokines or a full 30+ panel, RAWsphere ensures your multiplex data is accurate and reproducible.
Fluidics-Free Technology
EYRA is built around a completely flow-free design. No sheath fluid, priming, waste handling or blocked probes.
Instead of pushing samples through fluidics, EYRA uses confocal microscopy to image settled magnetic beads directly in the wells of a 96-well plate. Each bead carries a unique fluorescent dye signature for identification, while analyte-bound PE-labelled antibodies provide the quantitative signal.
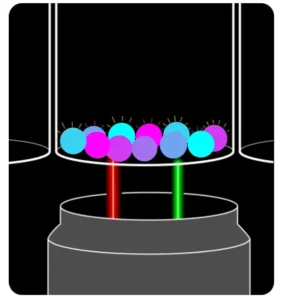
The result? You insert your plate, select your assay in the intuitive Mabtech Opal™ software, and hit read. Fifteen minutes later, you have fully processed data – with your samples never leaving the wells.
From Plate to Excel - Fast
Opal™ software comes preloaded with templates for every EYRAplex kit. Standards, plate layouts, and gating are handled automatically – so you spend less time setting up and more time on results.
Once the plate read is complete, results are exported directly into Excel, with options for bulk export if you’re working on large studies or multiple plates. No manual reformatting. No data wrangling headaches.
The Maintenance-Free Mindset
Because EYRA is fluidics-free, there’s no daily calibration or cleaning. No flushing, no wasted consumables, and no time lost to instrument downtime. It’s genuinely a plug-and-play experience – switch it on, run your plate, and walk away with your data.
Joe’s Takeaway
The Mabtech EYRA isn’t just another multiplex platform – it’s multiplexing reimagined. By removing fluidics and harnessing high-resolution confocal imaging, EYRA delivers:
- High-plex capacity: 30+ analytes per well
- Fast turnaround: ~15 minutes per plate
- No maintenance: no daily cleaning or calibration
- Accurate, reproducible results: powered by RAWsphere analysis
For labs looking to streamline their workflow without sacrificing data quality, EYRA offers a fresh, frustration-free alternative to traditional flow-based systems.
Until next time… happy experimenting!
Joe Roberts, PhD
Product Manager
Millennium Science

Spot the Difference: ELISA vs ELISpot vs FluoroSpot
A Joe Blogs post by Joe Roberts, PhD
When it comes to measuring proteins such as cytokines, antibodies, or growth factors, there’s no shortage of options. ELISA, ELISpot, and FluoroSpot all have important roles to play. While each assay relies on antibody-based detection, knowing which one to use (and when) can mean the difference between simply collecting data and gaining real insight.
As a Product Manager at Millennium Science, I speak with researchers every day who are deciding between these techniques. In this blog we will explore each assay to help you spot the difference and help you choose the right assay for your next experiment.
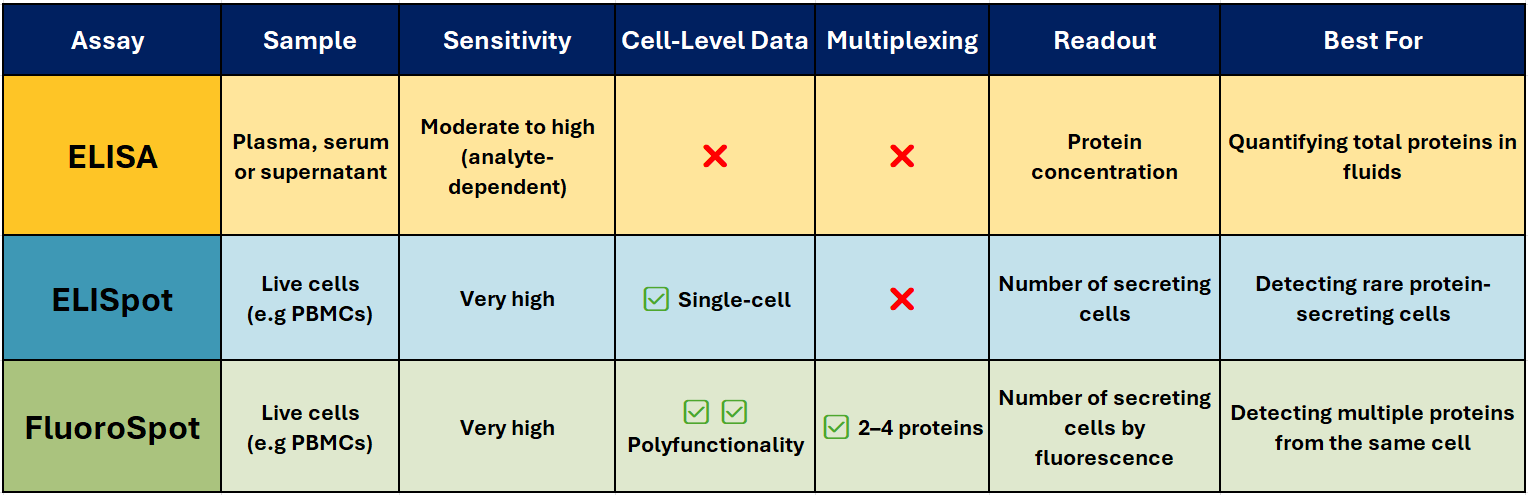
ELISA – The Trusted Workhorse 📊
Best for: Quantifying total soluble protein in biological fluids.
ELISA (Enzyme-Linked Immunosorbent Assay) is one of the most widely used immunoassays for detecting and quantifying soluble proteins, such as cytokines, antibodies, and hormones, in serum, plasma, or cell culture supernatants. It’s robust, scalable, and ideal for high-throughput analysis when you need accurate, reproducible concentration data.
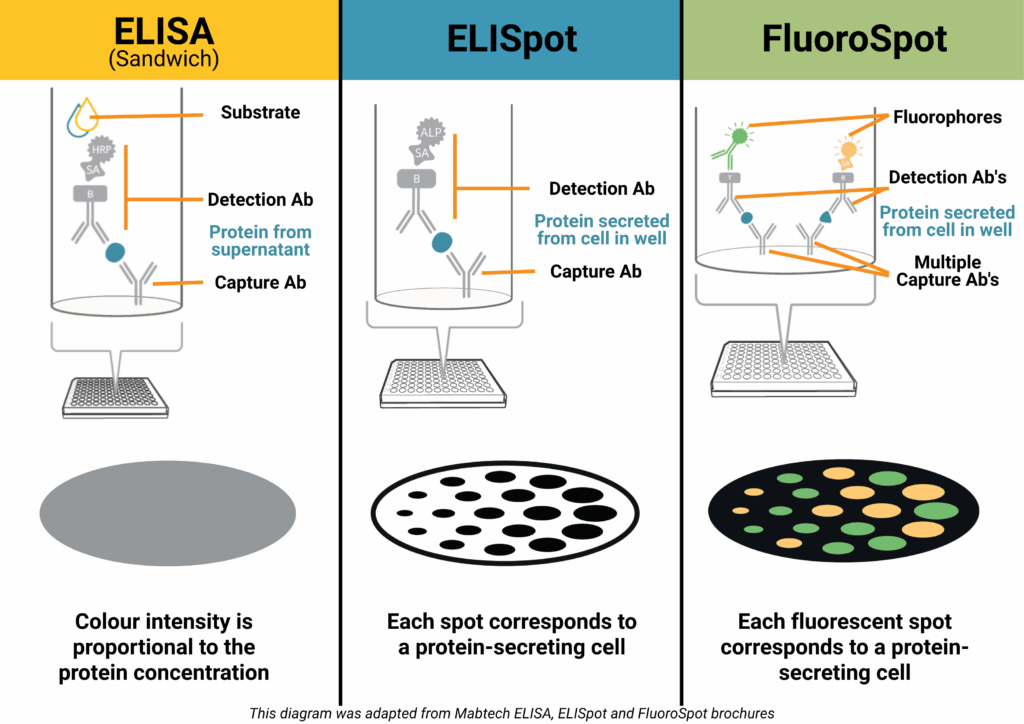
How it works (Sandwich ELISA):
A capture antibody is coated onto a high-binding plate and binds the target protein in your sample. A biotinylated detection antibody binds a different epitope, followed by a streptavidin–enzyme conjugate. Addition of a colourimetric substrate produces a measurable signal proportional to protein concentration.
Why researchers choose it:
- Delivers quantitative results (e.g., pg/ml or ng/ml)
- Compatible with high-throughput and automation
- Well-established, with widely available equipment
- Ideal for comparative analysis across multiple samples
Limitations:
- No information on the number or type of cells producing the protein
- Sensitivity can be lower than single-cell assays
ELISpot – Spotlight on Secreting cells
Best for: Counting individual protein-secreting cells.
ELISpot (Enzyme-Linked ImmunoSpot) detects and counts cells that secrete a specific protein. It’s particularly valuable when studying immune responses where the frequency of antigen-specific T or B cells is low – for example in vaccine research, oncology, and autoimmune disease studies.
How it works:
Live immune cells are added to a PVDF plate pre-coated with a capture antibody. When stimulated, the cells secrete the target protein, which is immediately bound near the cell. After washing away cells, a detection antibody and enzyme conjugate are added. A precipitating substrate forms a visible spot at each secretion site – each spot representing a single responding cell.
Why it stands out:
- Extremely sensitive – can detect one responder cell in >100,000
- Functional readout of immune activity
- Ideal for rare antigen-specific responses
- Widely used in T-cell response monitoring, vaccine trials, allergy research, and immuno-oncology
Pairing with Mabtech’s ASTOR2 automated ELISpot reader delivers rapid, high-precision spot counting with consistent, reproducible results across entire assay plates, and when used with Mabtech monoclonal antibody pairs and ready-to-use ELISpot kits, ensures reliable performance across species and sample types.
FluoroSpot – Multiplex Made Easy 🌈
Best for: Analysing polyfunctional immune responses.
FluoroSpot builds on the ELISpot principle but uses fluorescently labelled detection antibodies to identify multiple proteins secreted by the same cell – typically 2-4 proteins. This allows you to measure not just whether a cell responds, but how it responds.
Why it’s powerful:
- Detects polyfunctionality – cells secreting multiple cytokines
- High sensitivity with low background
- Enables simultaneous detection of different functional subsets
- Excellent for vaccine research, infectious disease, and cancer immunotherapy
Pairing with Mabtech’s IRIS2 ELISpot, FluoroSpot and FociSpot reader enables precise, high-resolution spot detection across multiple fluorescence channels.
Joe’s Takeaway
- ELISA → When you want to know how much protein is present in a sample.
- ELISpot → When you need to know how many cells are producing a protein.
- FluoroSpot → When you need to know which protein a cell is producing, and whether it’s producing more than one at the same time.
In many projects, using ELISA and ELISpot/FluoroSpot together can give the clearest picture — combining quantitative bulk measurements with functional single-cell insights.
If you’d like help selecting the right assay for your research, get in touch with our team at Millennium Science. We’re local experts, and Mabtech’s range covers everything from research use to clinical-grade applications.
Until next time… happy experimenting!
Joe Roberts, PhD
Product Manager
Millennium Science

Welcome to Joe Blogs
A Fortnightly Take on Life Science Technology 🔬
Hi, I’m Joe Roberts, PhD – Product Manager here at Millennium Science, immunologist at heart, and someone who’s passionate about helping researchers get the most out of their tools, assays and experimental design. My career has taken me through PhD, post-doctoral, and senior scientist roles, including roles within flow cytometry core facilities across the UK, Ireland and Australia. That journey has shaped a deep appreciation for the technologies that enable great science.
Here at Millennium Science, our mission is to ignite life science discovery with revolutionary technology!
This blog series – which I’m calling Joe Blogs – is my way of sharing insights, practical tips, and a few behind-the-scenes stories from the technologies I work with every day. From immunoassays and cell-based analysis to real-time monitoring and flow cytometry, I’ll be unpacking the platforms and reagents that drive discovery across Australia and New Zealand’s life science community.
What You Can Expect from Joe Blogs ✍️
Whether you’re running your first experiment on a new platform or refining an advanced workflow, Joe Blogs is designed to help you:
✅ Understand what your instruments can (and can’t) do
✅ Compare assay options with clarity and context
✅ Get practical insights from someone who’s been there
✅ Stay on top of emerging techniques and technologies
Think of it as a conversation, where we talk shop about the platforms you’re using, the questions you’re asking, and the science you’re working to advance.
Joe Blogs – Upcoming Topics 📅
Here’s a taste of what’s in the pipeline:
- Spot the Difference: ELISpot vs FluoroSpot vs ELISA
- Real-Time Results: What xCELLigence Really Tells You About Cell Health
- NovoCyte or NovoCyte Opteon? Choosing the Right Flow Cytometer
- Setting Up Your First ELISpot Assay: Tips for Success from Day One
I’d Love to Hear from You 💬
Got a burning question, a technical challenge, or a topic you’d love to see unpacked in a future post?
Let me know! I’d really like this blog to reflect the real conversations and challenges happening across our local research community.
Whether it’s about assay design, data interpretation, instrument comparisons or workflow optimisation, your ideas will help shape future editions of Joe Blogs. You can reach me directly via email or through the team at Millennium Science.
Let’s Keep It Real (and Local) 🤝
As part of Millennium Science, we’re proud to partner with leading life science suppliers to bring world-class tools to Australian and New Zealand researchers, with the local support to match. My goal with Joe Blogs is to give you a bit of that support upfront: advice, clarity, and maybe even a shortcut or two.
So, whether you’re measuring T cell responses, analysing cell proliferation, or exploring new approaches to data-rich experiments, you’re in the right place.
Until next time…happy experimenting!